Chapter 7: Synthetic Biology — The Design–Build–Test–Learn Loop for Efficacy Compounds
Why This Chapter
(Chapter 4) and (Chapter 5) showed how candidate molecules get narrowed. This chapter takes the next question — how do you make those molecules by the ton? Gram-scale isolates from nature do not run a cosmetic industry. A single SK-II line consumes hundreds of tons of fermentation filtrate per year, and L'Oréal's tens of thousands of global SKUs each demand tens to hundreds of ppm of active ingredient. The distance from "we found it in nature" (discovery) to "industry can use it" (production) — even after AI has narrowed the candidate set — is the territory synthetic biology fills.
The single clearest sentence Brief B asked the book to address sits here — "the domain of predicting which efficacy compounds a microbe can make, and then producing those compounds at scale." This chapter maps the tools, industrial cases, and regulatory coordinates of that workflow.
Three quantitative anchors for this chapter 1. The DBTL (Design–Build–Test–Learn) loop has become the standard work unit of cosmetic synthetic biology. [18] gave the first templating case with artemisinic acid in yeast; [16] scaled it from mg/L to 25 g/L; [1] is the current AI-accelerated DBTL playbook. 2. CRISPR-Cas9, base editing, and prime editing have collapsed the cost of strain engineering on industrial chassis (E. coli, S. cerevisiae, Y. lipolytica) by an order of magnitude. [6] published mouse-gut in situ base editing in Nature — one of the first demonstrations of editing a live commensal in vivo, and a leading indicator of the regulatory collision that live cosmetic microbes are about to face. 3. Industrial coordinates: Amyris (squalane commercialization → 2023 bankruptcy → 2024 ingredient-focused restructuring [2]), Ginkgo Bioworks (platform model [12]), Zymergen (2021 IPO → effectively wound down by 2022), Geltor (2025 PrimaColl FDA GRAS letter — first ingestible regulatory clearance for biodesigned collagen [11]), Evonik × Modern Meadow (Vecollage Fortify L — vegan collagen commercially launched for cosmetics [10]). Live engineered cosmetic microbes have no regulatory home — the political core of this chapter (Gap 5).
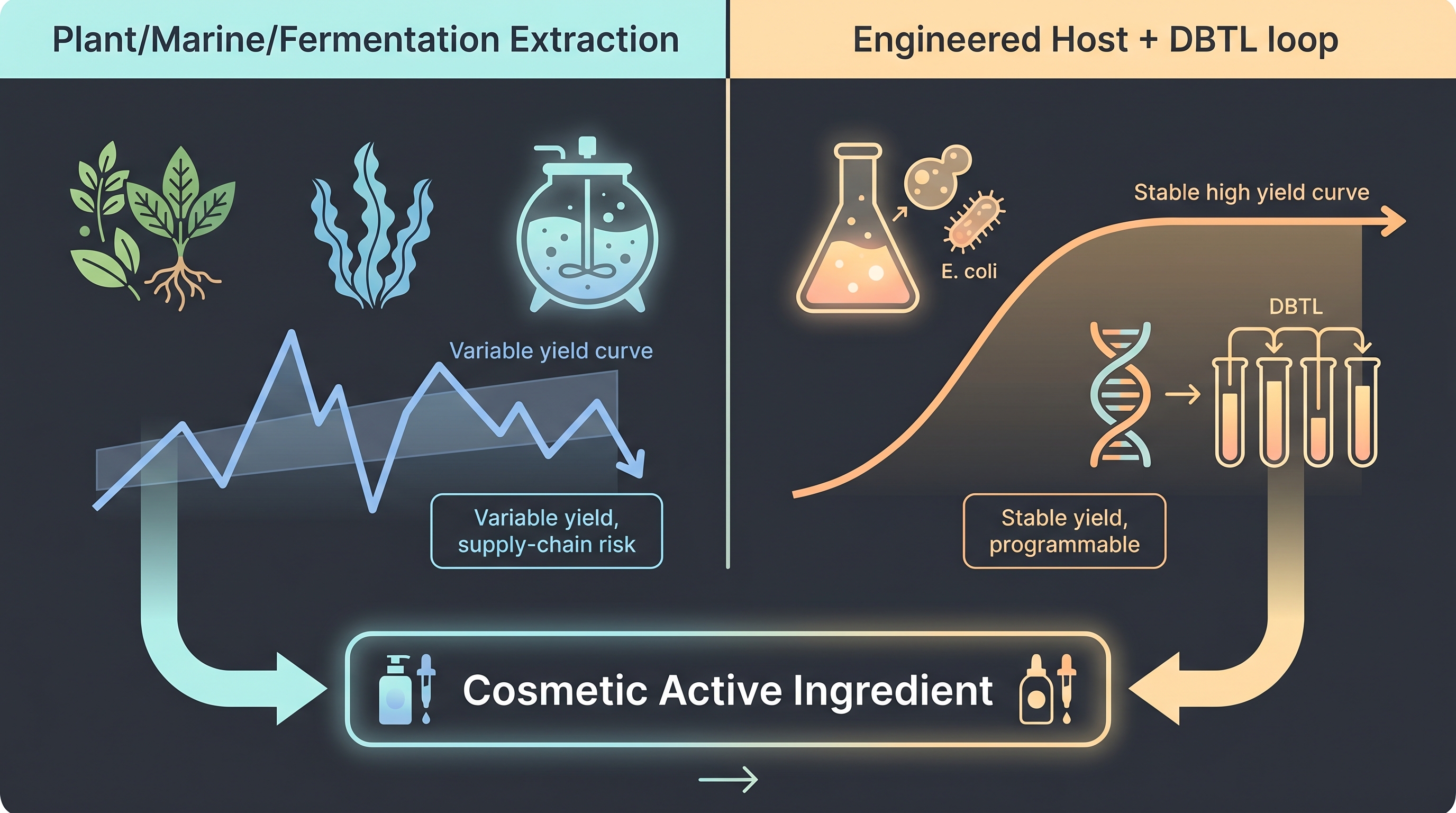
7.1 From Extract to Engineered Host — Why "We Found It in Nature" Isn't Enough
The first generation of cosmetic actives relied on extraction. Isolate a compound from a plant, marine source, or fermentation product; validate efficacy of that isolate; ship it. The limits of this model — to borrow Brief B's framing — show up simultaneously on three axes: yield, seasonality, and stability.
Yield ceilings. The most-cited case is artemisinin. The natural source plant Artemisia annua contains only 0.01–1.1% artemisinin by dry leaf weight; one hectare of cultivation yields roughly 1–5 kg. Meeting tens of tons of global demand required thousands of hectares of coordinated cropland across India, China, and East Africa. That structure was the direct motivation for the [18] Nature paper — transplanting the plant mevalonate pathway into Saccharomyces cerevisiae to produce artemisinic acid microbially.
The cosmetic analog is squalane. Natural squalane was historically extracted from shark liver oil, with industry estimates of ~500 g per shark and roughly 2.7 million sharks landed annually to meet global ton-scale demand. Once Amyris began producing the same molecule from S. cerevisiae fermentation — and later via Neossance Hemisqualane / the Aprinnova joint venture — shark-derived squalane was effectively retired at cosmetic grade [2]. The same pattern is expanding into vegan collagen (Geltor, Modern Meadow), plant aromatics (Ginkgo Bioworks), and peptide actives.
Seasonality and stability. Plant extraction is exposed to harvest and year-to-year variability. An engineered fermentation host, once established, can produce the same molecule at the same titer on a 365-day batch cadence. That difference is the real reason industry is migrating from extraction to engineered hosts.
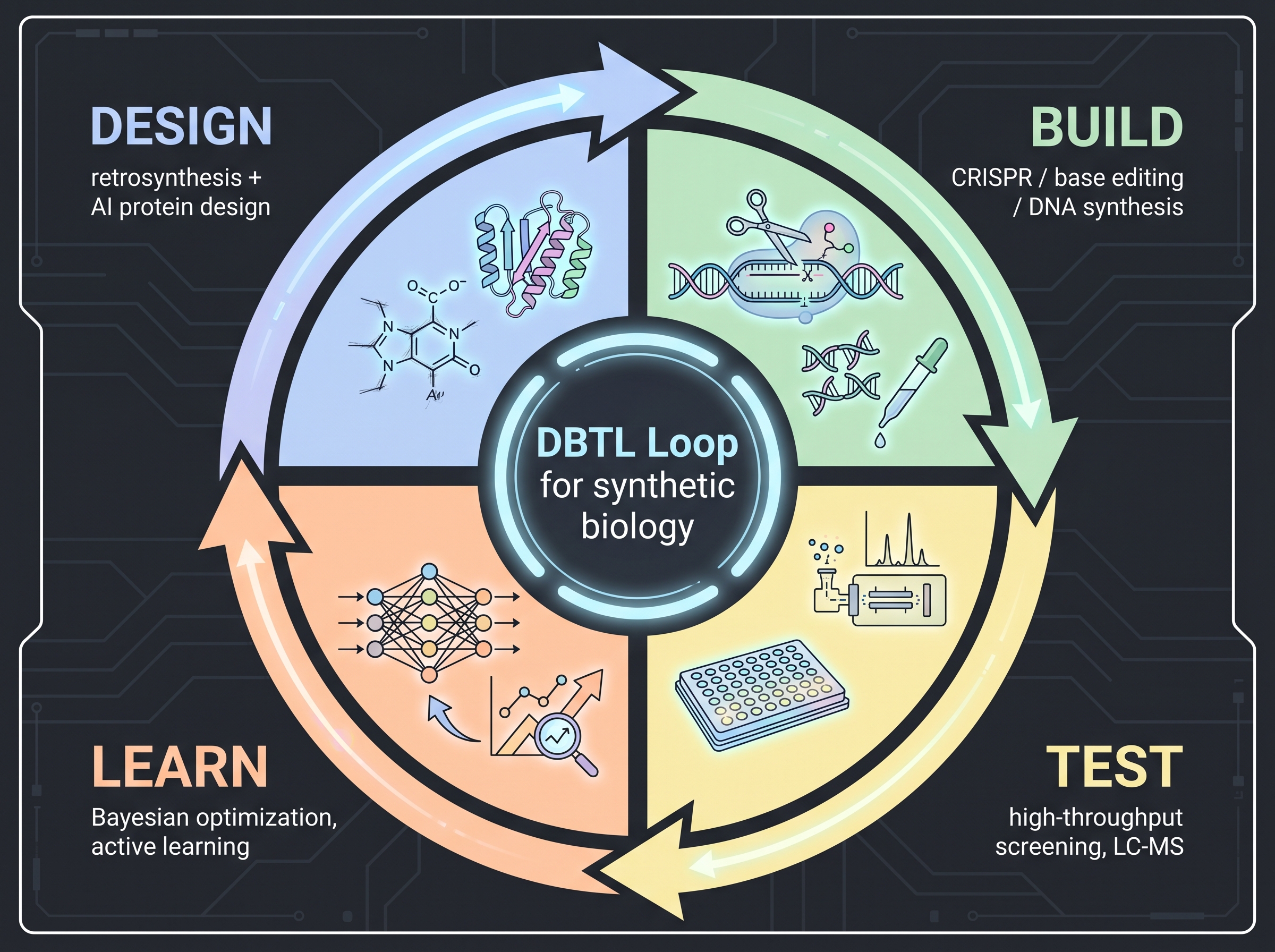
7.2 The DBTL Loop — The Standard Work Unit of Synthetic Biology
DBTL (Design–Build–Test–Learn) is the de facto cycle of cosmetic synthetic biology [1]. What used to take 18–36 months per turn now compresses to 6–12 months with AI assistance. Step by step:
Design — computational pathway design and retrosynthesis. Once a target molecule is fixed (a vegan collagen peptide, an AHR-agonist metabolite, an anti-aging spermidine derivative), the next question is which enzymes from which host should be expressed in what order. Two streams compete.
- Retrosynthesis AI — start from the target molecule and search backward through known enzymatic steps to propose feasible synthetic routes. MIT's ASKCOS, AstraZeneca + Chalmers' AiZynthFinder, and Allen AI/Cambridge's Chemformer are the leading tools. Chemical retrosynthesis and enzymatic retrosynthesis are distinct domains, but since 2024 the two tool families have increasingly fused with enzyme databases (BRENDA, KEGG, MetaCyc) for cosmetic and food natural-product route proposal.
- Protein design models — as seen in (Chapter 5) (Profluent's ProGen2 [19], Cradle Bio, and so on) design new enzyme variants. Retrosynthesis proposes "what reactions are needed"; protein-design models propose "what enzyme variant performs that reaction."
Build — DNA synthesis and strain engineering. Twist Bioscience, IDT, Ansa Biotechnologies, and other DNA synthesis providers materialize the designed pathway, which is then integrated into the host. Integration methods have evolved rapidly.
- CRISPR-Cas9 knock-in / knock-out — the standard tool for precise insertion into industrial chassis (E. coli, S. cerevisiae, Y. lipolytica, P. putida).
- Base editing (cytidine / adenine deaminase) — fine-tune enzyme variant activity by single-base substitution. After settling in vitro, [6] published mouse-gut in situ base editing in Nature, demonstrating feasibility in live hosts — the technical foundation for the live-cosmetic-microbe regulatory debate (Gap 5).
- Prime editing — replaces longer sequences more precisely than base editing. Still being standardized for cosmetic fermentation hosts.
- OpenCRISPR-1 [19] — Profluent's ProGen2 protein language model trained on the universe of CRISPR-Cas proteins to generate new genome editors. Some variants were released open-source — about 400M parameters, deployable on industrial chassis — accelerating the commoditization of cosmetic synthetic biology tools.
Test — high-throughput screening. From hundreds to tens of thousands of synthesized strain variants, measure (1) whether the strain is alive (growth rate), (2) whether it makes the target molecule (LC-MS / HPLC), and (3) whether it makes enough (g/L titer). Fluorescent reporters, biosensors, FACS, and droplet microfluidics operate at successive stages. Where (Chapter 4)'s ML triage narrowed molecular candidates, this stage narrows strain candidates.
Learn — ML over the data. Each cycle's results feed back into the next Design phase. Bayesian optimization, active learning, and Gaussian process regression are the standard tools. Ginkgo Bioworks' SSI (Selections and Strain Improvement) program [12] is the industrial form of this learn phase — fortified by the 2022 acquisition of Altar (adaptive lab evolution), combining in vivo evolution with ML-guided design.
7.3 Strain Engineering Tools — Specificity by Industrial Chassis
The industrial chassis most commonly used in cosmetic synthetic biology are E. coli (cheap, fast growth, proteins and peptide actives), S. cerevisiae (eukaryotic PTMs, terpene natural products, squalane), Y. lipolytica (lipid-based molecules, sphingolipids, ceramides), and P. putida (tolerance to toxic intermediates, polymer monomers). Each chassis has distinct CRISPR-Cas9 efficiencies and base-editing constraints, so pathway allocation during retrosynthesis is decided together with chassis selection.
A host the cosmetic industry watches closely is Y. lipolytica — many core cosmetic molecules are lipid-based (squalane, ceramide, fatty acid esters) and Y. lipolytica natively accumulates lipids. For the same reason, since around 2022 multiple reports show Y. lipolytica outperforming S. cerevisiae on squalane titer.
Engineering live commensals sits on a different axis. Rather than extracting molecules from a fermentate, this approach engineers S. epidermidis, C. acnes, or Lactobacillus directly so the live strain is applied to skin and produces the active on the skin. Leading cases:
- S-Biomedic — engineered C. acnes expressing RoxP [20]. RoxP is a redox protein that absorbs radicals under oxidative stress, with a clinical hypothesis around acne and erythema. Beiersdorf took a majority stake in 2022 [5] and folded the asset into its Microbiome Design Platform at the 2024 Capital Markets Day.
- Eligo Bioscience — CRISPR-Cas-armed bacteriophages that selectively eliminate or edit specific C. acnes strains [9]. Patent EP4240843A1.
- Parallel Health — a phage cocktail targeting inflammaging and redness [17].
- Arcaea — a workaround approach that designs prebiotics (microbial nutrient molecules) rather than living microbes [3]. Lowest regulatory burden.
These four cases are the lead candidates in [4]'s Cosmetics review of the bioengineered skin microbiome, and the first industrial cohort of live cosmetic microbes. None of the three live-microbe cases has secured a regulatory path for launching the live form — the subject of the next section.
7.4 Retrosynthesis AI — A Proposal Engine for Cosmetic Natural-Product Routes
Retrosynthesis tools enter the cosmetic domain along two tracks.
First, chemical retrosynthesis. ASKCOS, AiZynthFinder, and Chemformer were built for small-molecule pharma. Applied to cosmetic natural products (kojic acid, salicylic acid derivatives, AHR-agonist ligands), they propose chemical step sequences but often include steps that no in vivo enzyme can run, or routes that don't align with enzyme propensities. The usage pattern is hybrid — view the chemical route, then translate into an enzymatic alternative.
Second, enzymatic retrosynthesis. Apply the same retrosynthesis framework against enzyme databases (BRENDA, MetaCyc), restricting each step to known enzymatic reactions. RetroPath (the Toulouse group), Selenzyme, and BioNavi are academic exemplars. Industrially, Ginkgo Bioworks and Inscripta have disclosed internal variants.
Retrosynthesis becomes the natural interface for the molecular design outputs of (Chapter 4) — when a generative model emits "this molecule satisfies endpoint X," retrosynthesis answers "and here is how to synthesize/produce it," handing the result to the Design step of the DBTL loop. The fusion of the two tool families entered industrialization in 2024.
7.5 The Industrial Canon — Five Cases, Five Lessons
① Artemisinin in yeast (2006 → 2013) — the first templating case. [18] inserted the plant mevalonate pathway, amorphadiene synthase, and amorphadiene oxidase into S. cerevisiae to produce the artemisinin precursor artemisinic acid at the mg/L scale — the first Nature publication of this workflow. Seven years later, [16] scaled the same workflow to 25 g/L, and via the Amyris × Sanofi partnership became a WHO-recognized industrial line. That seven-year gap (mg/L → 25 g/L) reveals the actual time scale of synthetic biology industrialization. The biggest bet in the field now is that AI-accelerated DBTL can halve that gap.
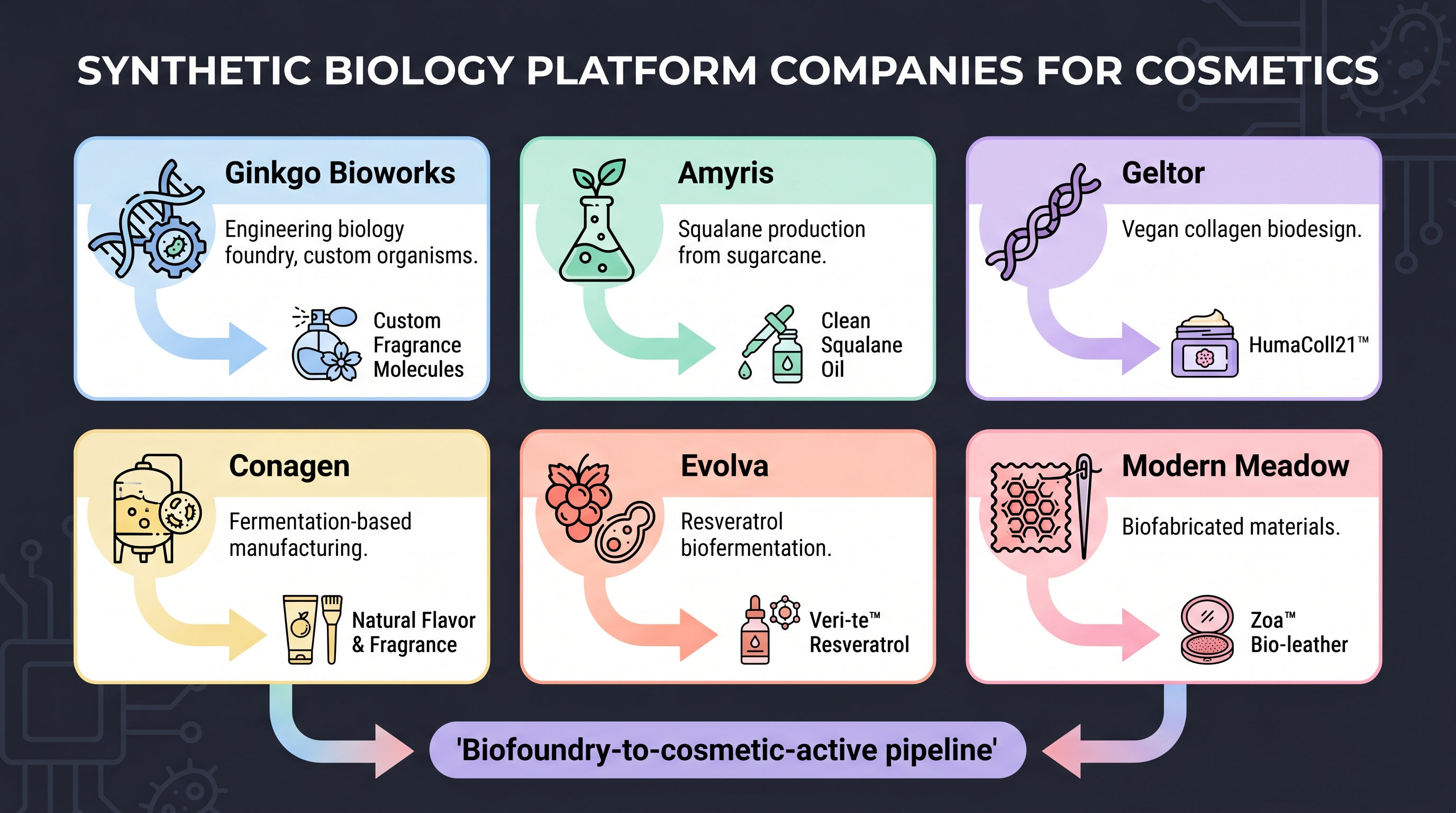
② Amyris squalane — the first commercial exit into cosmetics. Amyris commercialized squalane on the same yeast platform, entering the global cosmetic ingredient market. Via Neossance Hemisqualane and the B2C brand Biossance, it became the standard squalane ingredient for dozens of global skincare lines. But in August 2023 Amyris filed Chapter 11 — the commercial model (B2C brand expansion) had failed to support the capital intensity. After 2024 restructuring, the company refocused on ingredient manufacturing [2]. Lesson: the synthetic-biology ingredient molecule survived, but the company's business model did not.
③ Ginkgo Bioworks — the platform model. Ginkgo does not own molecule IP directly; it takes cosmetic, fragrance, and food companies on as customers and engineers strains to their spec. Since the 2021 SPAC listing the share price has fallen more than 95%, but the business model is intact, and the 2022 Altar (adaptive lab evolution) acquisition and 2024 SSI program restructuring [12] are expanding cosmetic and personal-care partnerships. Lesson: a platform model without molecular IP takes longer to validate capital efficiency than an IP-owning model.
④ Zymergen — the cautionary tale. Zymergen pitched ML-driven strain engineering at its 2021 IPO. But the failure of product-market fit for its first product, Hyaline (a transparent polymer film), combined with insufficient technical validation, drove a 76% share-price collapse in the same year; Ginkgo effectively absorbed the company in a fire-sale acquisition in 2022. Zymergen never directly entered cosmetics, but its IPO-and-collapse story is now the canonical reference for the risk profile of the synbio IPO model. Lesson: invoking AI/ML alone does not shorten the industrialization timescale of synthetic biology — the molecule has to fit a real market.
⑤ Geltor PrimaColl + Evonik × Modern Meadow Vecollage — vegan collagen industrialization. Geltor designs and produces vegan collagen, replacing animal-sourced collagen via microbial expression. In October 2025 [11] received an FDA GRAS ("no questions") letter for PrimaColl, an ingestible collagen peptide — the first ingestible regulatory clearance for biodesigned collagen. In the same category, Evonik × Modern Meadow commercially launched Vecollage Fortify L — a vegan collagen for cosmetic personal care — in February 2024 [10]. Lesson: this is the first cosmetic synthetic-biology active category to travel from "discovery" all the way to "regulatory clearance + commercial launch." Time scale: roughly ten years (Modern Meadow founded 2011 → Vecollage launched 2024).
7.6 Cosmetic Applications — Coordinates by Category
Vegan collagen / elastin / silk. Geltor PrimaColl (ingestible [11]), the Geltor cosmetic line (microbially expressed collagen), Modern Meadow × Evonik Vecollage Fortify L ([10]), and Bolt Threads (microbial silk protein). Product-market fit for this category is durable — driven by BSE / porcine-prion concerns over animal-extracted collagen, vegan/halal certification demand, and molecular uniformity.
Fermented peptides (Korean industry). KOLMAR Korea, COSMAX BTI, and Amorepacific produce active peptides via fermentation from their internal strain collections. [14]'s Communications Biology paper on spermidine-mediated dermal structural recovery is the academic readout from that Korean industrial line; [15]'s EPI-7 postbiotic RCT (8-week clinical) is covered in detail in (Chapter 9). COSMAX's 2nd-generation platform [7] and the HelloBiome partnership [8] are the follow-on industrialization layer.
Engineered Lactobacillus / Cutibacterium / Staphylococcus. Engineering live commensals to produce target metabolites on the skin rather than via fermentate extraction. S-Biomedic's RoxP-expressing C. acnes [20] and Eligo's CRISPR phages [9] are the leading examples. But — see the next section — the regulatory route for live launch is unresolved.
Recombinant proteins (growth factors, recombinant elastin / silk). Cosmetic use of growth factors like EGF, FGF, and IGF has been commoditized by animal-cell or microbial expression. Korean R&D is strong in this category, but peer-reviewed efficacy and safety readouts at cosmetic grade remain sparse (Gap 1).
7.7 The Regulatory Frontier (Gap 5) — The Vacuum Around Live Engineered Cosmetic Microbes
This is the chapter's most political content. [4] is explicit — "GMO regulatory frameworks (FDA, EU CTR) are not designed for live cosmetic microbes; safety and ethical guidelines lag synthetic biology's capabilities."
Mapping the coordinates:
- FDA OTC cosmetic monograph — assessment framework presupposes stable chemical formulations. Self-replicating organisms are not in the taxonomy.
- EU CTR (Cosmetic Product Regulation 1223/2009) — Annex assessment items also lack a living-microorganism category. S-Biomedic and Eligo are exploring EU launch routes but no published framework exists.
- Korea MFDS Functional Cosmetics — the same vacuum. Freeze-dried heat-killed strains and cell-free fermentates (postbiotics) can pass under existing categories, but live commensals remain unresolved.
- FDA GRAS for ingestible biosynthetic ingredients — [11]'s PrimaColl is the first to clear it. But it covers an ingestible collagen — not applicable to live engineered microbes in topical form.
- FDA-approved oral microbiome drug — [21]'s VOWST (SER-109) became the first FDA-approved drug for C. difficile recurrence prevention. The first live microbe cleared through the pharmaceutical path — not a route a cosmetic company can pursue commercially, but a regulatory signal that live microbes can clear a high safety bar.
The industry has narrowed its workarounds to two:
- Postbiotics / heat-killed strains — do not apply the strain itself; either extract only the molecules produced by fermentation, or apply the strain after killing it. KOLMAR, COSMAX, and Amorepacific all rely on this workaround. Lowest regulatory burden.
- Live commensals — the form S-Biomedic, Eligo, Parallel Health, and Arcaea are attempting. The regulatory route is as of May 2026, not formally established in any jurisdiction.
[6]'s Nature paper on mouse-gut in situ base editing demonstrated that live commensals can be edited in vivo, foreshadowing the largest regulatory flashpoint cosmetic industry will face in the next five years. [13]'s early synthesis of horizontal gene transfer risk — the lateral spread of antibiotic resistance genes through the human microbiome — is a natural starting point for regulatory analysis. Ordinary cosmetic users have daily contact between hand and face microbiomes and any live engineered strain applied to skin, so four checks must run simultaneously — GMO classification, environmental exposure, user consent, and traceability. This chapter sets up the starting point of that review; (Chapter 12) integrates it into a policy blueprint.
7.8 Signals from Adjacent Industries — What Pharma and Food Synthetic-Biology Industrialization Teach
Given the cosmetic industry's regulatory vacuum, precedents from adjacent industries matter. (Chapter 11) does the full analysis; here are the key coordinates.
- Pharma — live microbe: [21] VOWST. Demonstrates that a live microbe can reach FDA drug-approval grade.
- Food — synthetic-biology ingredient: [11] PrimaColl. Microbially expressed collagen peptide cleared ingestible GRAS.
- AI drug discovery's clinical readout: From [22]'s GENTRL DDR1 inhibitor in Nature Biotechnology through Insilico Medicine's rentosertib Phase 2a, the ability of AI-designed molecules to reach clinical readouts has been validated. The cosmetic industry has had no equivalent readout (Gap 1). The first clinical readout for a cosmetic active produced via DBTL is most likely to come from Korean industry (COSMAX, Amorepacific, LG H&H) or a mid-tier EU firm (S-Biomedic, Eligo).
7.9 Open Questions
- The new baseline DBTL timescale — How far can AI-accelerated DBTL compress the mg/L → g/L titer journey below the seven-year baseline of the [16] era? An externally verified timeline for any specific cosmetic active molecule does not yet exist.
- First regulatory clearance for live cosmetic microbes — Which of FDA, EU CTR, or Korea MFDS will publish the first framework? Where do the precedents of VOWST apply to cosmetics, and where do they break down?
- Monitoring horizontal gene transfer — Among the risks enumerated by [13], which must be reviewed first for cosmetic topical application? Environmental persistence, household transmission, and use during pregnancy or breastfeeding each need a scenario-specific assessment framework.
- Molecular IP vs platform IP — Amyris's molecule-IP model led to Chapter 11; Ginkgo's platform-IP model has shed 95% of its share price. Which model offers higher capital efficiency for a cosmetic synbio startup? No validated answer exists.
- User perception and vegan certification vs synbio labeling — Synthetic-biology ingredients clear vegan certification easily, but a subset of consumers avoid "GMO-derived" labels. How will cosmetic labeling and communication standards evolve over the next five years?
References
- ACS Synthetic Biology editorial collective (2024). Synthetic Biology of Natural Products Engineering — DBTL cycle advances. ACS Synthetic Biology, 2024. [ACS Synth. Biol., 2024]
- Amyris Inc. (2024). Amyris post-bankruptcy restructuring — return to ingredient manufacturing for cosmetics. BeautyMatter / Cosmetics Business / Green Chemicals Blog, 2024. [Amyris, 2024]
- Arcaea (2024). Arcaea precision prebiotics for armpit and scalp microbiome modulation. Arcaea patent WO2024118975A1; HPC Today feature 2025. [Arcaea, 2024]
- Atallah, C., El Abiad, A., El Abiad, M. et al. (2025). Bioengineered Skin Microbiome: The Next Frontier in Personalized Cosmetics. Cosmetics 12(5):205.
- Beiersdorf AG (2022). Beiersdorf majority stake in S-Biomedic — Microbiome Design Platform integration. Beiersdorf press 2022 acquisition + 2024 Capital Markets Day strategy update. [Beiersdorf, 2022]
- Brödel, A. K., Charpenay, L., Galtier, M. et al. (2024). In situ targeted base editing of bacteria in the mouse gut. Nature, May 2024.
- COSMAX USA (2024). COSMAX 2nd-Generation Skin Microbiome platform + Microbiome AI. Global Cosmetic Industry, 2024. [COSMAX, 2024]
- COSMAX BTI and HelloBiome (2025). COSMAX × HelloBiome microbiome-powered personalized skin/scalp care. WWD / Personal Care Insights, 2025. [COSMAX & HelloBiome, 2025]
- Eligo Bioscience (2024). Eligo Bioscience CRISPR-Cas-armed phage for C. acnes. Eligo patent EP4240843A1; Atallah 2025 review citation. [Eligo, 2024]
- Evonik and Modern Meadow (2024). Evonik launches biotech-based vegan collagen ingredient for the cosmetic industry (Vecollage Fortify L). Evonik press, Feb 2024. [Evonik & Modern Meadow, 2024]
- Geltor Inc. (2025). Geltor PrimaColl — first FDA GRAS letter for biodesigned vegan collagen. FoodNavigator-USA / SynBioBeta, Oct 2025. [Geltor, 2025]
- Ginkgo Bioworks (2024). Engineering at scale: an intro to Selections and Strain Improvement (SSI). Ginkgo Bioworks blog + Altar acquisition (2022), Feb 2024. [Ginkgo, 2024]
- Huddleston, J. R. (2014). Horizontal gene transfer in the human gastrointestinal tract: potential spread of antibiotic resistance genes. Infection and Drug Resistance, 2014.
- Kim, G., Kim, M., Kim, M. et al. (2021). Spermidine-induced recovery of human dermal structure and barrier function by skin microbiome. Communications Biology 4:231.
- Kim, J., Lee, Y. I., Mun, S. et al. (2023). Efficacy and Safety of Epidermidibacterium Keratini EPI-7 Derived Postbiotics in Skin Aging: A Prospective Clinical Study. International Journal of Molecular Sciences 24(5):4634.
- Paddon, C. J., Westfall, P. J., Pitera, D. J. et al. (2013). High-level semi-synthetic production of the potent antimalarial artemisinin. Nature 496, 528–532.
- Parallel Health (2024). Parallel Health phage cocktail for skin inflammaging and redness. Parallel Health product page + HPC Today 2025. [Parallel Health, 2024]
- Ro, D.-K., Paradise, E. M., Ouellet, M. et al. (2006). Production of the antimalarial drug precursor artemisinic acid in engineered yeast. Nature 440, 940–943.
- Ruffolo, J. A., Nayfach, S., Gallagher, J. et al. (2024). Design of highly functional genome editors by modeling the universe of CRISPR-Cas proteins (Profluent ProGen2 / OpenCRISPR-1). bioRxiv 2024.04.22.590591.
- S-Biomedic (2024). S-Biomedic engineered C. acnes RoxP-expressing probiotic strain. S-Biomedic press / HPC Today 2025; Atallah 2025 review citation. [S-Biomedic, 2024]
- Seres Therapeutics and Nestlé Health Science (2024). VOWST (SER-109) commercial trajectory — first FDA-approved oral microbiome therapeutic. FDA label + commercial updates, 2024. [Seres, 2024]
- Zhavoronkov, A., Ivanenkov, Y. A., Aliper, A. et al. (2019). Deep learning enables rapid identification of potent DDR1 kinase inhibitors. Nature Biotechnology 37, 1038–1040.