Chapter 12: A Blueprint for New Research — Data, Regulation, Reproducibility, and Opportunity
Why this chapter
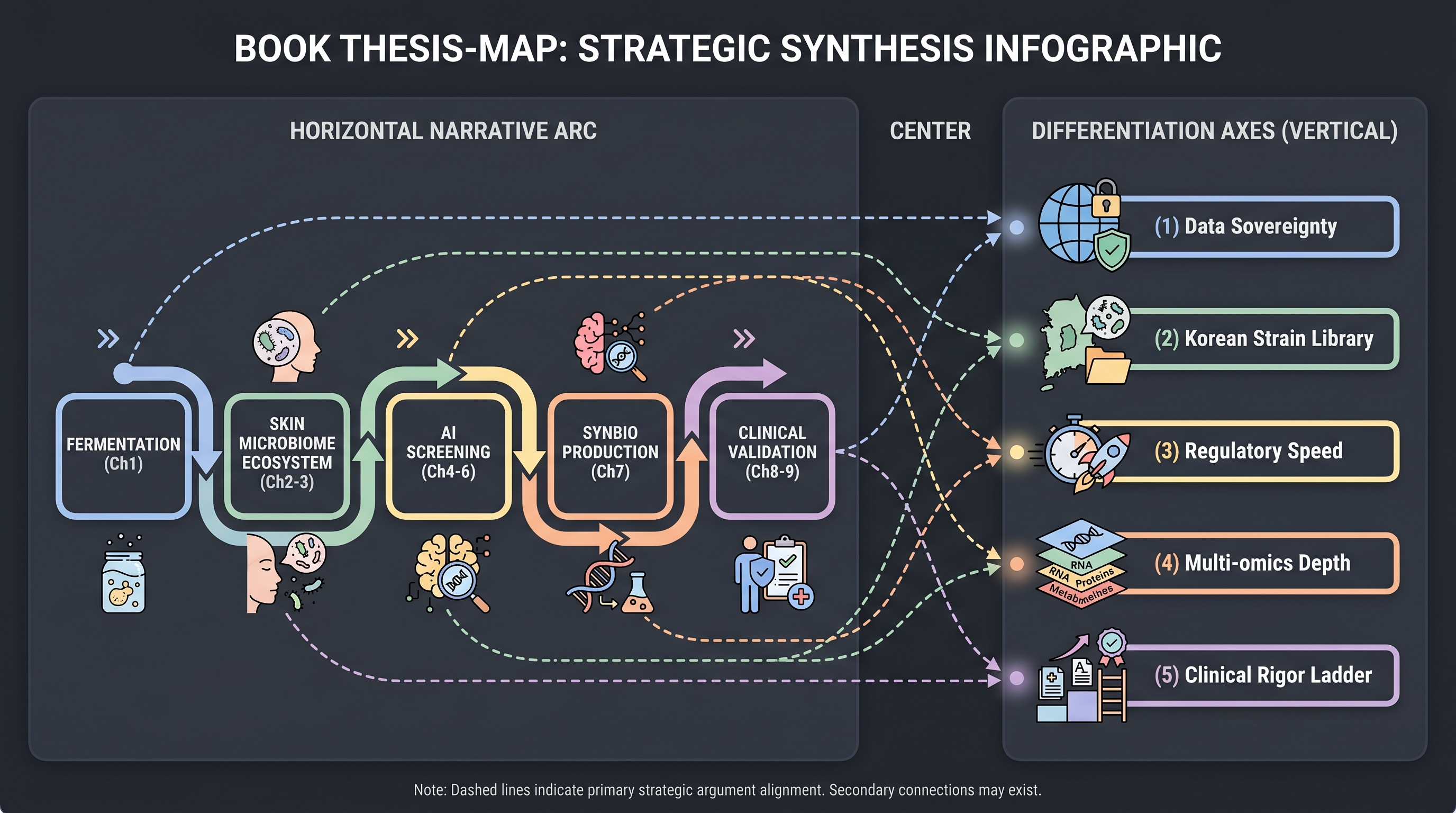
This book has traced a single arc across eleven chapters — fermentation → microbiome ecosystem → AI acceleration → synthetic-biology production → clinical validation. This closing chapter compresses that arc into a decision frame an R&D planner, a biotech founder, a regulator, or a journalist can take to their desk tomorrow. That is exactly what Brief B asked for — "ideas to apply new technologies in planning new research." It is also what Brief A asked for less directly — the question "is fundamental research now obsolete?" was always a stand-in for "where should I deploy my budget next year?"
This chapter is unlike two adjacent text genres. It is not a comprehensive review — the kind that ends with "more multi-omics studies are needed" [17]. It is not a consulting deck either — the kind whose six-month-horizon recommendations rarely survive contact with reality. Instead, it maps the book's eight chapters of evidenced gap diagnoses (15 entries in gaps.md) and eleven chapters of industry case mapping onto a 12-to-60-month decision matrix. The book's gap inventory and competitor mapping are the load-bearing inputs.
Three quantitative facts to anchor this chapter 1. AI toolkit is commodity since 2024; the cosmetic-clinical readout vacuum is intact as of May 2026. AlphaFold3, Boltz-1, Chai-1, ESM3, and RoseTTAFold-All-Atom were all released in 2024 [1]. Yet zero peer-reviewed clinical readouts of AI-designed cosmetic actives exist (Gap 1) [5]. This asymmetry is the single largest opportunity vector of the next five years. 2. Data asymmetry is structural. Unilever holds an in-house cohort of ~5 billion data points (30,000 samples); L'Oréal and COSMAX hold comparable internal corpora [40]. By contrast, the only meaningful public skin-microbiome reference catalog is iHSMGC — 10.9M genes from a Chinese cohort, with approximately 45% reported as East-Asian-specific novel genes relative to HMP [27]. No skin-microbiome foundation model analogous to ESM3 (98B parameters, 7B-parameter weights open) [18] exists today (Gap 2 + Gap 6). 3. Korea is a first-class region, but published visibility concentrates in one cluster. The COSMAX-Dankook-HuNBiome axis has produced the FACE-LINK 950-subject cohort [28], the EPI-7 clinical trial [23], and the spermidine recovery mechanism work [22] — all peer-reviewed. Amorepacific and LG H&H — the two largest Korean cosmetic firms by revenue — are absent from peer-reviewed channels [2]. The asymmetry itself is analytic content.
12.1 The book's central thesis, in one page
The book's eleven chapters compress into five load-bearing claims. They are the foundation of every decision frame that follows, so they are restated here in their tightest form.
1. Fundamental research is not obsolete — it is the ground truth AI rests on. (Chapter 3, Chapter 9). The answer to Brief A's "are bacterial culture, isolation, and basic antimicrobial/anti-inflammatory testing now outdated?" is no, they are complementary. The Gallo-Nakatsuji bacteriotherapy lineage [29] reached autologous Phase 1 without AI — pure culture + AMP assay + immunology. The molecular mechanism of Galactomyces ferment filtrate [39] was filled in retroactively — three decades after SK-II's Pitera was commercialized. AI's contribution is clear, but the cycles it compresses still close at the wet-lab bench.
2. The AI inflection has a precise vintage and a precise topology. (Chapter 4, Chapter 5). 2021 is "year zero" — AlphaFold2 [21], RoseTTAFold [6], iHSMGC [27], Carrieri-Unilever XAI [8], COSMAX-spermidine [22], ShA9 Phase 1 [30], and ZOE PREDICT 1 [4] all converge in a single year. 2024–2026 commoditizes the toolkit — AF3 → Boltz-1 → Chai-1 → ESM3. The cosmetic industry has had AI-design stacks as a commodity for two years and has not yet published a single clinical readout. That vacuum is the opportunity vector (Gap 1).
3. The AI–clinical gap is not a cosmetic-internal limitation — adjacent industries have already closed it. (Chapter 9, Chapter 11). Insilico Medicine's rentosertib (TNIK inhibitor, AI-discovered target + molecule) reached IND in 30 months and reported a Phase 2a result of +98.4 mL FVC vs −20.3 mL placebo over 12 weeks [35]. The operational template exists. Cosmetic actives do not need to clear the pharma evidentiary bar — but precisely because they do not, the first AI-designed cosmetic active to publish a peer-reviewed clinical readout will produce a market signal larger than any pharma readout.
4. Synthetic biology will redefine cosmetic ingredients faster than regulation can follow. (Chapter 7, Chapter 11). Live engineered cosmetic microbes — S-Biomedic's RoxP-expressing C. acnes [36], Eligo's CRISPR-armed phage [12], Arcaea's precision prebiotics [3], Parallel Health's phage cocktail [32] — are all entering market without a regulatory home. Geltor PrimaColl's GRAS letter [15] is a precedent for ingestible synbio ingredients; no topical-live precedent exists. Seres VOWST [37] is a drug precedent that cosmetic channels cannot commercially route through.
5. Korean industrial-academic clusters are a first-class asset, with publication asymmetry. (Chapter 10, Chapter 11). The COSMAX-Dankook-HuNBiome-Yonsei-SNU axis is the only non-Western end-to-end industry-academic pipeline publishing at international cadence in peer-reviewed channels. The combination of OEM channel (COSMAX) + academic anchor (Dankook) + strain resource (HuNBiome) is a structural advantage no Western competitor can replicate in the short term.
No existing source agrees with all five claims simultaneously. The D1–D5 analysis in positioning.md is the warrant.
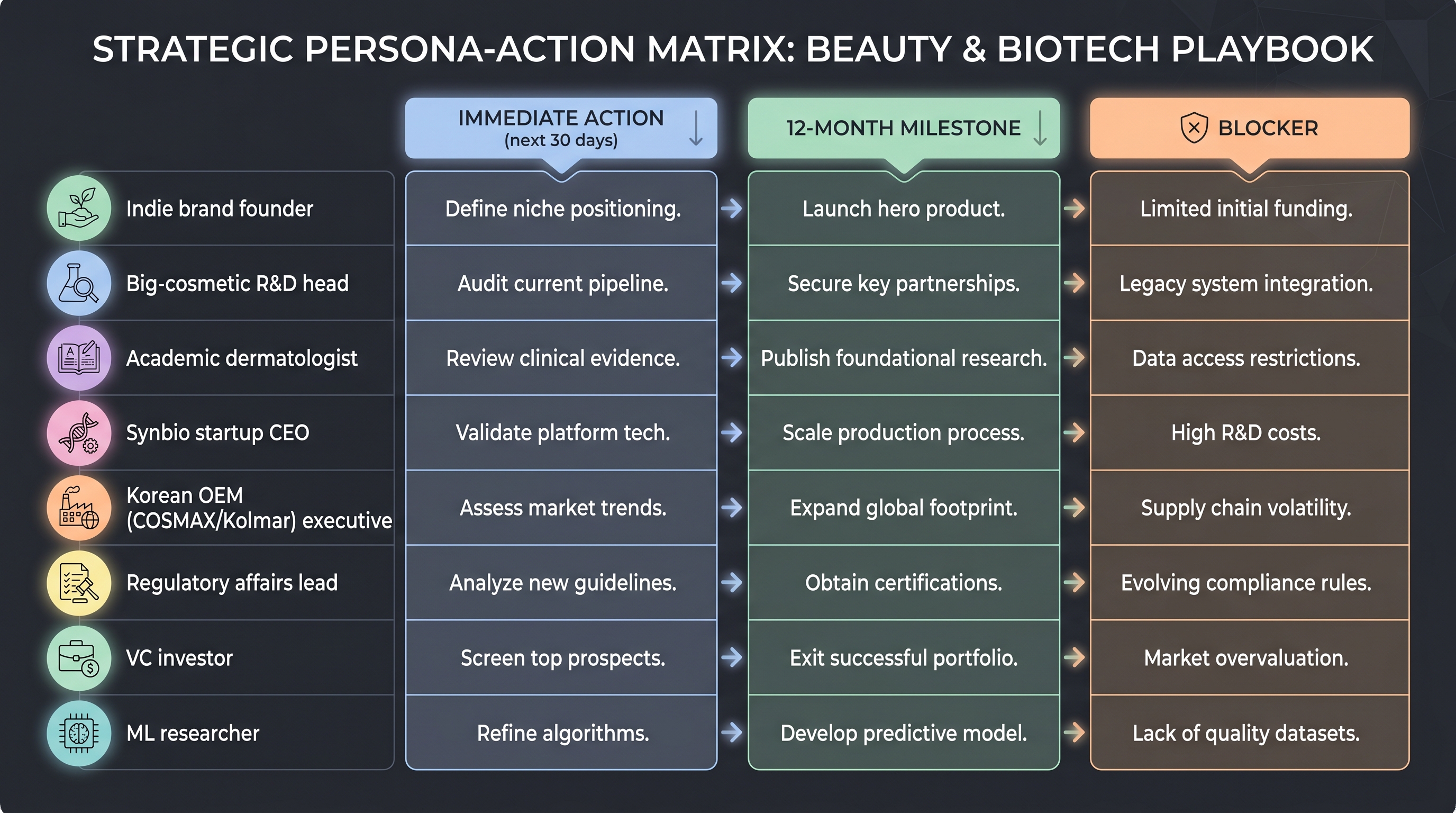
12.2 Decision frame by reader persona — the next 12 months
positioning.md defines eight reader personas the book explicitly targets. From the same conclusions, what to do tomorrow differs by persona. This section maps each persona to a single paragraph: "if you are X, the next 12 months should look like Y." This is the most operational part of Chapter 12.
Non-expert curious about the fermentation-to-data shift (Brief A reader). Target for the next 12 months: be able to grade microbiome cosmetic marketing claims on four axes — species-level vs strain-level, prebiotic vs probiotic vs postbiotic, ferment filtrate vs live engineered, and AI-generated claim vs peer-reviewed evidence. Chapter 2's strain-level resolution material (the C. acnes RT4/RT5 vs RT6 divergence [13]) and Chapter 7's synthetic-biology taxonomy give the vocabulary. Concrete recommendation: in a retail aisle, identify in one glance whether a microbiome product is strain-level, species-level, or just a ferment filtrate. If marketing text claims "AI-formulated," check whether a peer-reviewed publication is cited — as of 2026, zero exist, so almost every such claim is marketing-grade (Gap 15).
Korean R&D planner (Amorepacific, LG H&H, COSMAX, or a mid-tier brand) — the longest persona treatment. Target: align the in-house R&D portfolio either with the COSMAX-Dankook publication cadence or explicitly declare an IP-first, publication-avoidant strategy. The two paths require different KPIs. The publication strategy needs (i) a sustained academic co-authorship with Dankook, Yonsei, or SNU; (ii) a one-year commitment to a cohort at FACE-LINK 950-subject scale [28] or larger; (iii) adopting the Chapter 9 clinical-simulation/ex-vivo pipeline as in-house ground truth. The IP-first strategy needs (i) a robust KIPRIS-filed patent landscape; (ii) a trade-fair cadence of press-grade industry disclosures (Amorepacific / LG H&H style); (iii) external audit-ready calibration data backing virtual-cohort claims (Chapter 8's "AI virtual cohort," "60% faster"). Either way, the decision itself is the first 12-month action. The book's diagnosis: Korea's OEM channel + academic axis is an asset Western competitors cannot replicate in four to six years if and only if a publication strategy is chosen (Gap 4).
Cosmetic R&D scientist inside a global major (L'Oréal, Unilever, Estée Lauder, Shiseido). Target: expose one auditable surface of the internal AI stack to an external (academic or regulatory) collaborator within 12 months. The cleanest candidate is a single Carrieri-2021-style SHAP-attributed gradient-boosted regressor [8] committed to a peer-reviewed channel. Reason: Gap 15 (no independent evaluation of industry-grade AI claim-generation pipelines) is the only blocker that can be closed voluntarily before the expected 2027 FTC / EU CTR scrutiny tightens, and the first mover defines the next-five-year industry standard. Secondary recommendation: against Chapter 4's buy-the-data pattern, explicitly grade in-house strain × phenotype × formula × clinical-mapping data as the first-class asset — ranked above model architecture.
Biotech / synbio founder considering the cosmetic adjacency. Target: price the regulatory entry cost of cosmetic market entry explicitly. Pattern from Chapters 7 and 11: the live-engineered-microbe path has no regulatory home, so short-term (≤5 years) market entry is difficult. The two immediately viable paths are (i) synbio-produced ingredient (Geltor-PrimaColl style — GRAS-letter eligible, B2B channel [15]); (ii) prebiotic-only (Arcaea-style [3] — efficacy claims without a live organism). The book's view: whether to pursue cosmetic and pharma channels in parallel is a cap-table decision. Whether Insilico's potential cosmetic-active spin-out (unconfirmed as of book release) materializes will test whether cosmetics can be a credible "second product" for an AI-pharma firm.
Pharma scientist considering microbiome cosmetic adjacencies. Target: an explicit decision on whether the in-house microbiome program treats cosmetic adjacency as a portfolio element or rejects it. Book's observation (Gap 11): big pharma (Pfizer, Novo Nordisk, GSK) has no public microbiome headline programs — Pfizer × XtalPi [33] and Novo × Cradle [44] are AI partnerships, not microbiome headlines. The likely cause is the Seres-VOWST commercial setback, which has dampened public disclosure. Cosmetic adjacency may emerge in the next 12 months as a fewer-regulatory-hurdles route to ROI on microbiome IP.
Journalist / analyst covering the microbiome cosmetic industry. Target: internalize one of the five load-bearing claims as a frame for reporting. The most under-reported frames: the Korean cluster (Gap 4) and live-engineered regulation (Gap 5). The most over-covered frame: AI design itself — whether a model gets smarter matters less than whether a clinical readout gets published.
Regulatory / policy professional (FDA, EU CTR, MFDS). Target: document two regulatory vacuums — (i) live engineered cosmetic microbes (Chapter 7 Gap 5) and (ii) AI-derived efficacy claims (Chapter 8 Gap 15). Both are likely to become salient around 2027 driven by industry events (Eligo / S-Biomedic product launches; FTC guidance on AI claims). The cleanest anchors are the Geltor GRAS letter [15] (ingestible precedent) and Seres VOWST [37] (drug precedent).
AI researcher considering microbiome as a target domain. Target: map the minimum viable dataset coalition for a skin-microbiome foundation model (Gap 2). Unilever 30K, COSMAX 1K (FACE-LINK), and L'Oréal proprietary data, federated, are the only path to ESM3-scale corpora. The book's view: federated learning over a data cooperative is the most under-explored instrument at the academia-industry interface today.
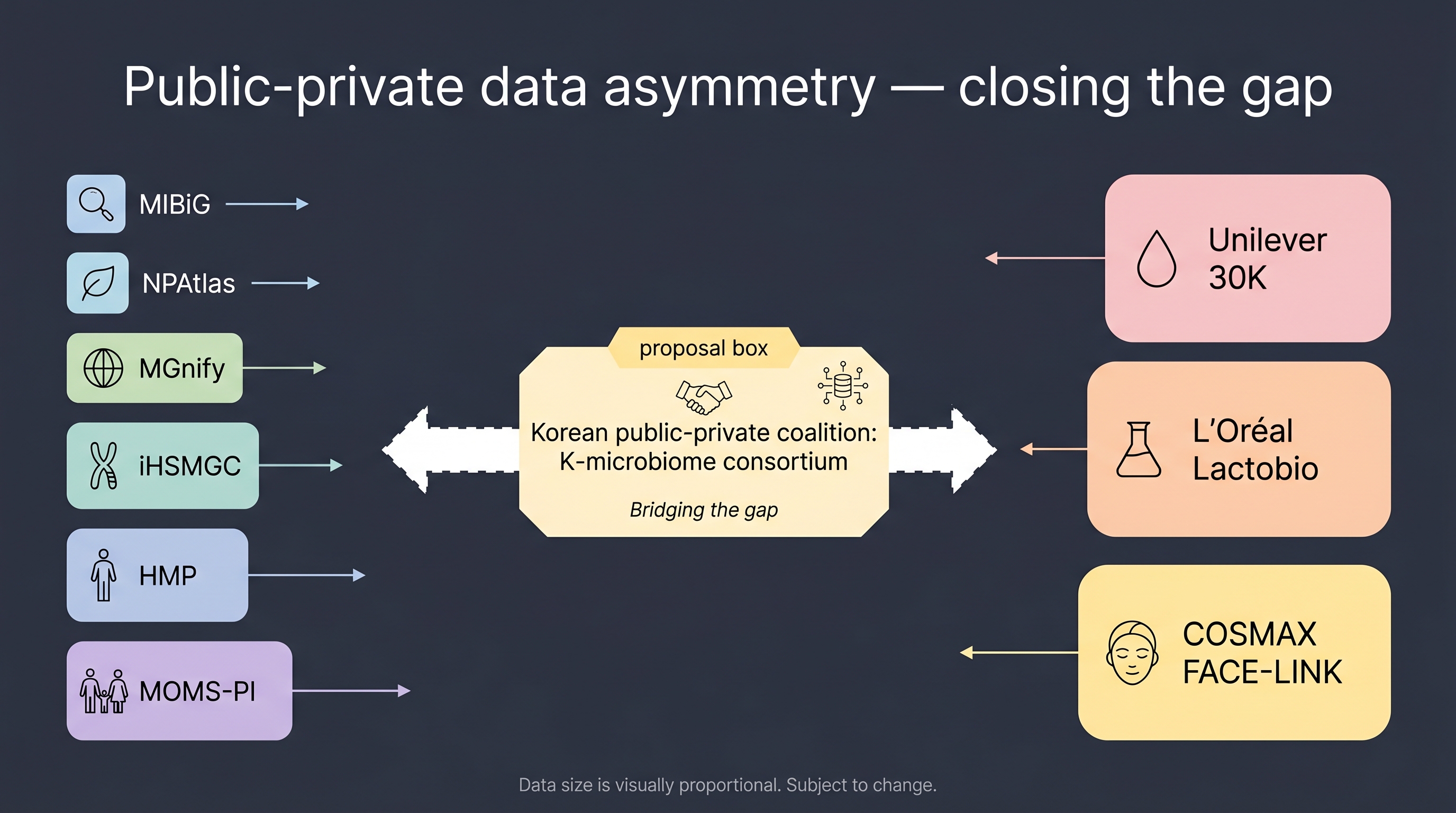
12.3 The data bottleneck — open vs proprietary, and a Korean public cohort proposal
Data asymmetry is the field's deepest single constraint. ESM3 was possible because UniProt + AlphaFold DB are public assets; cosmetic microbiome has no analog.
12.3.1 The asymmetry, anatomized
Three resource classes exist.
Public infrastructure. MGnify (EBI metagenome) is the largest public archive of environmental and human metagenomes, but skin coverage is roughly an order of magnitude smaller than gut. MIBiG (Minimum Information about a Biosynthetic Gene cluster) makes efficacy metabolite candidates traceable to biosynthetic gene clusters and is peer-reviewed robust. NPAtlas (Natural Product Atlas) is a peer-reviewed inventory of natural-product chemistry. iHSMGC [27] is the largest published skin-microbiome reference gene catalog to date. With ~10.9 million genes from a Chinese cohort and approximately 45% reported as East-Asian-specific novel genes relative to HMP, it makes two facts unavoidable: (a) reference catalogs are region-locked, and (b) global cosmetic-microbiome AI models generalize as their training data does. That is the start of the equity concern.
Semi-public consortia. The Human Microbiome Project (HMP) provides a NIH-led, ~250-subject skin cohort. MOMS-PI (Multi-Omic Microbiome Study: Pregnancy Initiative) is pregnancy/neonatal-focused with some skin sites. Neither is a cosmetic-efficacy benchmark — they offer composition only.
Proprietary. Unilever's 30,000-sample, ~5-billion-data-point internal cohort [40]. L'Oréal's Lactobio strain library plus internal cohort. COSMAX's FACE-LINK 950-subject cohort [28]. Combined, these three resources alone plausibly reach ESM3-training scale — but no single asset is publicly citable. Carrieri 2021 [8] (Unilever × IBM) is the only peer-reviewed exposure of internal-cohort data — n=62 plus a UK validation cohort.
12.3.2 A Korean public-private skin microbiome cohort proposal
The solution to the asymmetry above is a data coalition, and Korea has the structural conditions to build one.
Proposal skeleton. Four-way coalition — government (MFDS or KIST under NST) + industry (COSMAX, Amorepacific, LG H&H) + academia (Dankook, Yonsei, SNU) + clinical (Seoul National University Hospital, Severance, Samsung Medical Center dermatology). Scale target: 5,000–10,000 subjects over 2–3 years; FACE-LINK's 950-subject cohort [28] provides feasibility evidence. Open layer: composition (16S + partial shotgun) plus skin-phenotype data (hydration, TEWL, sebum, pH, age, Fitzpatrick), de-identified, with a two-year embargo before public release. Industry-private layer: each partner's formula-efficacy mapping stays internal — only peer-reviewed publications expose pieces. Governance: integrated IRB across three hospitals; data standardization to MIxS plus MOMS-PI compatibility; kit-bias mitigation via a single kit lot and a single sampling protocol (responding to the Chapter 3 reproducibility diagnosis).
The justification — why Korea? Compared with the US, EU, and China, Korea has (i) a single-language, unified-IRB system that accelerates cohort operations; (ii) an OEM-dominated industry structure (COSMAX) that uniquely minimizes IP conflict among competitors sharing a cohort; (iii) demographically East-Asian skin that is underrepresented in global references (HMP, iHSMGC aside); (iv) policy framing (K-Bio and K-Beauty) that already permits classifying such an industrial indicator as a national asset.
Cost estimate — 5,000 subjects, 2 years longitudinal, 16S + partial shotgun + standard phenotype + cross-link to clinical: approximately ₩5–8 billion (≈ USD 4–6M), shared between government and four firms. This is about one-fifth the scale of Unilever's 30K cohort, but it would be the largest East-Asian cohort accessible in peer-reviewed channels.
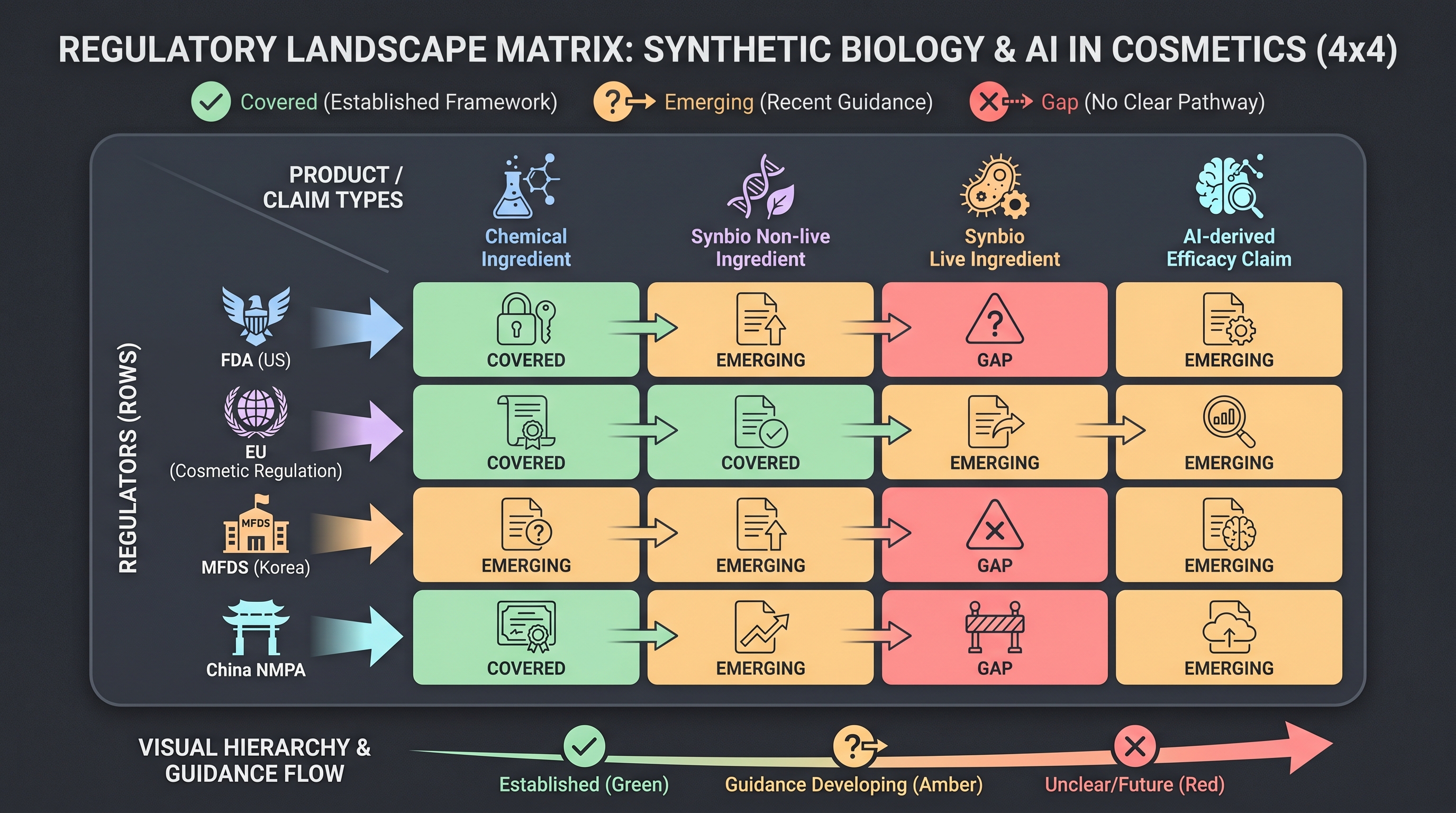
12.4 The regulatory frontier — FDA, EU CTR, MFDS, and the live engineered microbe
The four major cosmetic regulatory frameworks — FDA OTC cosmetic monograph + Cosmetic Modernization Reform Act (MoCRA, 2022), EU Cosmetic Regulation (EC 1223/2009) + Clinical Trials Regulation (CTR), MFDS Functional Cosmetic notification, and China NMPA Special Cosmetics — were all written for stable chemical formulations. Self-replicating biological entities and AI-derived efficacy claims have no clear home in any of them.
12.4.1 The two regulatory gaps
Gap 5 — Live engineered cosmetic microbes. S-Biomedic's RoxP-expressing C. acnes [36], Eligo's in-situ CRISPR base-editing phage [12], Arcaea's precision prebiotics [3], and Parallel Health's phage cocktail [32] do not fit cleanly into any regulatory category. Atallah et al. (2025) [5] is explicit: "GMO regulatory frameworks were not designed for live cosmetic microbes." Two workarounds are in play — (i) postbiotic (kill the organism before packaging — loses the live-microbe upside), (ii) prebiotic-only (avoid the live organism, Arcaea model). Both abandon the live-organism upside. FDA route: if a cosmetic makes a drug claim it is automatically a drug, so a Seres-VOWST [37] route is technically open, but trial cost breaks cosmetic ROI. EU route: CTR governs clinical trials, and cosmetics typically fall outside CTR; live engineered cosmetics sit at the intersection of consumer product safety regulation and biosafety regulation. MFDS route: live engineered microbes are not named in the Functional Cosmetic notification, leaving a vacuum that is plausibly the first to provoke explicit Korean rule-making around 2027.
Gap 15 — AI-derived efficacy claims. Unilever's "60% faster consumer insight, 75% faster claim generation" [40], L'Oréal's AI Skin Genius, and COSMAX's Microbiome AI platform [10] all generate efficacy claims through AI virtual cohorts or AI recommendation engines — and there is no external audit framework for those claims. Di Guardo et al. (2025) [11] is explicit: "no harmonized regulatory framework for AI-derived efficacy claims." Likely path — the FTC (US advertising truthfulness) or the EU Digital Services Act + EU AI Act (adopted 2024, phased enforcement through 2026) will be the first to scrutinize AI-generated cosmetic claims. MFDS is positioned as the world's most likely first mover on an AI cosmetic-claim audit framework, marketable as a K-Beauty export-quality signal.
12.4.2 Three plausible 5-year regulatory scenarios
Scenario A — Geltor precedent extension. The GRAS-letter route ([15], Geltor PrimaColl, October 2025) extends to non-live synbio cosmetic ingredients. FDA expands its GRAS evaluation framework to topical synbio ingredients; first case plausibly 2027–2028. This path closes only half of Gap 5 (the non-live half).
Scenario B — Seres precedent adapted to cosmetics. VOWST [37] established an FDA precedent for live microbiome therapeutics. A cosmetic firm reframes a cosmetic indication as a drug claim (e.g., atopic dermatitis, acne) and routes through FDA Biologics. This closes 100% of Gap 5 — but trial cost breaks cosmetic-scale ROI unless the firm is a pharma-cosmetic hybrid.
Scenario C — A new regulatory category. FDA / EU / MFDS create a new category — "Live Cosmetic Microorganism" or "Bio-Cosmetic Active." Cleanest resolution but slowest — FDA rulemaking takes 3–5 years, and EU CTR / Cosmetic Regulation amendments take longer. MFDS's Functional Cosmetic notification revision could plausibly be the world's fastest first mover.
12.5 The reproducibility crisis — batch effects, kit bias, and East-Asian representation
Reproducibility has an explicit academic consensus — Papoutsoglou et al. (2023) [31], the pan-European COST Action — but the cosmetic-industry AI claim-generation pipeline has not deployed it (Gap 8).
12.5.1 Three systematic reproducibility threats
Batch effects. Identical microbiome samples can yield different results on different sequencing runs. When a cosmetic cohort spans two years of different sequencing lots — Unilever's 30K cohort is in this regime — batch correction algorithms can synthesize part of the apparent efficacy signal. ComBat and MMUPHin are academic standards; industry pipelines do not disclose batch-correction methodology.
Kit bias. DNA extraction kits (Qiagen DNeasy vs ZymoBIOMICS vs MO BIO PowerSoil) systematically enrich different taxa. Skin's low-biomass, host-contaminated environment makes kit bias larger than for any other body site. Chapter 3 diagnosis: very few cosmetic firms publish cross-kit-validated cohort data.
Ethnic / regional representation. iHSMGC [27] reporting approximately 45% East-Asian-specific novel genes relative to HMP is the cleanest evidence. If an in-house cosmetic AI model trains only on US/EU cohorts, generalization to East-Asian or darker-phototype skin is not guaranteed. Haykal et al. (2025) [17] is explicit: "geographic diversity is limited, darker phototypes underrepresented." This asymmetry is the largest single equity concern and the largest single business risk — a global product launch may fail efficacy in underrepresented demographics.
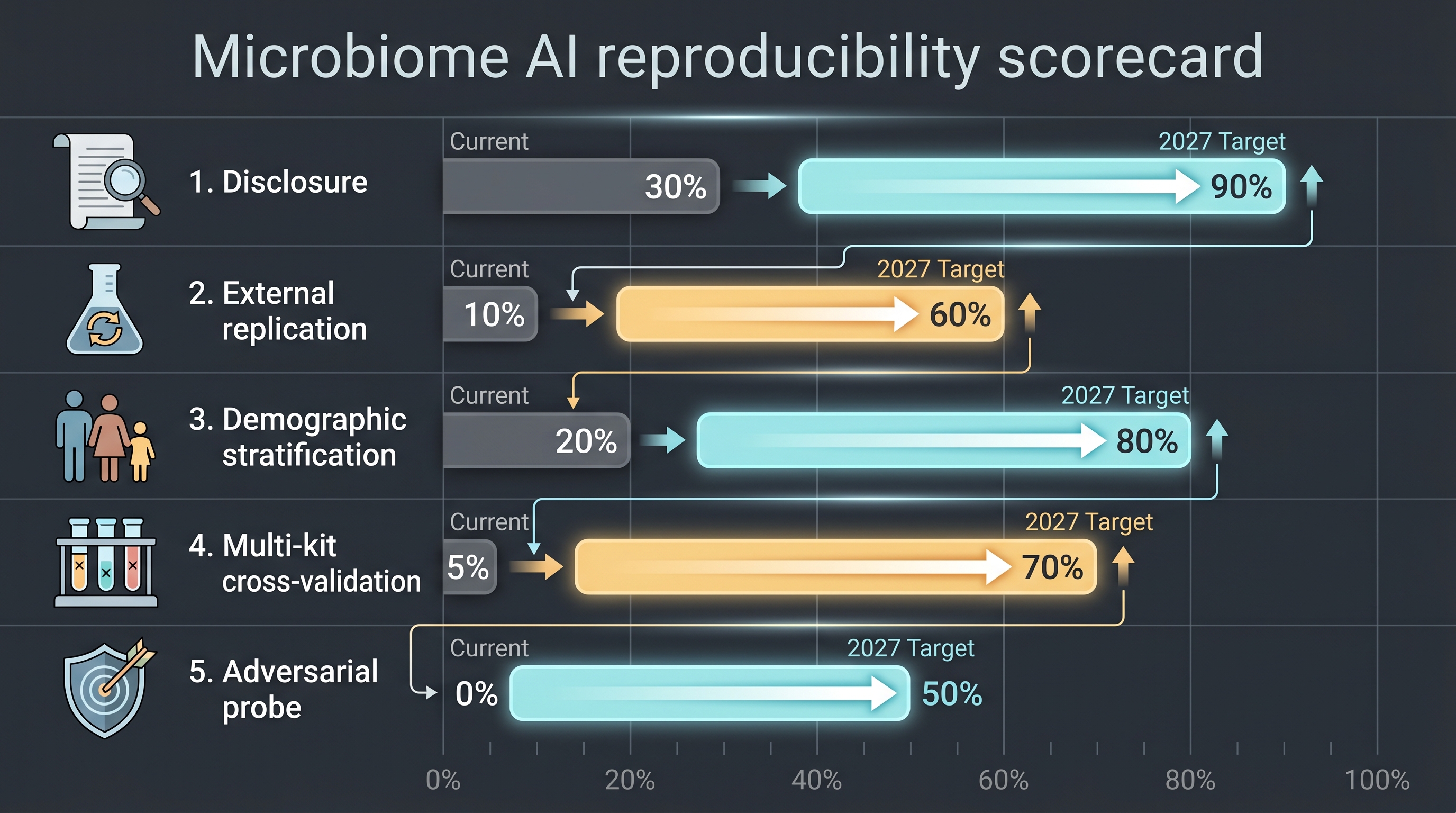
12.5.2 A cosmetic-industry adapted reproducibility checklist (proposal)
As a concrete contribution this book proposes a minimum viable reproducibility checklist, closing part of Gap 8:
- Standard disclosure layer — every cosmetic-microbiome AI claim must report cohort size, demographic split, kit, sequencing platform, and batch-correction method.
- External replication seed — deposit an anonymized 10–20% subsample of the internal cohort to a peer-reviewed channel so an external group can verify.
- Demographic stratification — report model performance separately for Fitzpatrick I–II, III–IV, and V–VI binarization.
- Multi-kit cross-validation — cross-validate 5–10% of the cohort with a second extraction kit.
- Adversarial probe set — red-team the AI claim generator with intentional-nonsense inputs (random microbiome compositions) and require it to abstain on those.
Combined with MIxS and MOMS-PI compliance — and assuming FTC / EU AI Act audits become mandatory around 2027 — cosmetic firms whose internal pipelines are already audit-ready will own first-mover advantage.
12.6 The foundation-model opportunity — honest anatomy of Gap 6
The most honest answer to Brief A's "why is AI an inflection?" is AlphaFold is an inflection for proteins; the microbiome inflection has not arrived yet. There is no ESM3 [18] (98B-parameter, 7B open-weight) or DNABERT-scale resource for skin-microbiome community function (Gap 2 + Gap 6).
12.6.1 What would be required?
Data scale. ESM3 was trained on ~2.7B protein sequences and ~236M structures. The microbiome analog, in (strain genome × condition × phenotype) tuples, plausibly requires ~10M tuples — public archives today (MGnify, NCBI combined) are at 100K–1M, one to two orders of magnitude short.
Compute scale. ESM3 training cost on the order of tens of millions of USD in GPU compute (~10–30M USD). A cosmetic-microbiome foundation model at that scale requires a cosmetic firm or government-grant + cloud-partnership arrangement.
Governance scale. The hardest piece. To assemble an ESM3-scale corpus, at least three of {Unilever, L'Oréal, COSMAX, NIH, BGI} must contribute to a common training corpus. Federated learning fits the use case — raw data stays on-premises and only trained gradients are shared. Industry incentive: if an aggregate model is meaningfully more powerful than any model trained on internal data alone — and if that difference translates into greater downstream ROI in cosmetic-active discovery — a coalition becomes rational.
12.6.2 First steps — what is possible within 12 months?
Three minimum viable steps:
Step A — pre-training corpus harmonization. Harmonize MGnify + iHSMGC + a slice of COSMAX FACE-LINK + Unilever's Carrieri-cohort exposure into a common schema (MIxS + MIMARKS) so the corpora become cross-referenceable. A one-year academic-consortium project.
Step B — task benchmark suite. Standardize skin-microbiome → efficacy-endpoint prediction tasks (hydration, TEWL, age, smoking — the Carrieri-2021 endpoint set [8]) with held-out test sets, baseline performances, and evaluation protocols. If academia adopts the tasks as common, model comparison becomes possible — the same mechanism that took ESM 1b → ESM2 → ESM3.
Step C — pre-trained encoder release. Release not a Carrieri-style gradient-boosted regressor but a Transformer encoder (DNABERT-architecture [20] or GeneBERT-style) that encodes 16S / shotgun composition into a latent representation. First buyers: academic cosmetic-microbiome labs. Largest users: mid-tier cosmetic firms without in-house AI infrastructure.
Sequentially or in parallel — and on a 2028–2030 timeline — these three steps put the first skin-microbiome foundation-model prototype within reach. Which nation and which firm becomes the main contributor is the most informative industry indicator to track over the next five years.
12.7 Five 5-year research bets — fundable, concrete, evaluable
This section compresses the book's eleven chapters of gap and case-study evidence into a five-year research portfolio. Each bet specifies (i) the gap it closes, (ii) success criteria, (iii) the most plausible host (academic, industry, or hybrid).
Bet 1 — A peer-reviewed clinical trial of an AI-designed cosmetic active (closes Gap 1)
Gap closed: Gap 1 — no peer-reviewed clinical readout of an AI-designed cosmetic active exists.
Core of the bet: take a cosmetic active (peptide or antioxidant) designed entirely in silico with AlphaFold3 / Boltz-1 / Chai-1 / ESM3 and run a Phase-2-equivalent randomized controlled trial with a peer-reviewed publication. Endpoint: TEWL, wrinkle volume, or spectrocolorimetry-quantified pigmentation showing a statistically significant difference vs placebo. Cohort size: ≥ 150 per arm, ≥ 12 weeks follow-up, double-blind.
Operational template: Insilico's rentosertib (target → IND in 30 months) [35]. Applying the same cycle time to a cosmetic active suggests design in 2026 → clinical readout in 2029.
Most plausible host: Korea (the COSMAX-Dankook axis, leveraging FACE-LINK [28]) or a mid-tier EU firm (Beiersdorf is a candidate). The Big-4 Western majors (L'Oréal, Unilever, Estée Lauder, Shiseido) follow IP-first strategies and are unlikely to publish — but competitive pressure could shift this by 2028–2030.
Bet 2 — A Korean public skin-microbiome cohort + benchmark (closes Gap 2 + Gap 12)
Gap closed: Gap 2 (no foundation-model-scale data) + Gap 12 (no open benchmark).
Core of the bet: the §12.3.2 proposal — Korean public-private cohort — plus a Carrieri-2021-style task suite [8] (hydration, TEWL, age, Fitzpatrick prediction) on that cohort, plus an annual public leaderboard, plus auditable inference for all submitted models.
Operational measurement: 2 years cohort collection → 1 year baseline benchmark → year 3 entry into a global academic + industry leaderboard (e.g., a NeurIPS or ICML workshop community challenge). By year 5, at least 10 external groups publish using the cohort.
Most plausible host: a four-way coalition of MFDS + NST + COSMAX + Amorepacific + LG H&H + Dankook / Yonsei / SNU. Korea's OEM structure uniquely minimizes IP conflict among competitors sharing the cohort.
Bet 3 — Skin culturomics × foundation model (closes Gap 3 + Gap 6)
Gap closed: Gap 3 (skin culturomics underdeveloped — gut culturomics is Lagier 2018 [25] but skin has no equivalent) + Gap 6 (no skin foundation model).
Core of the bet: transpose Lagier-style gut-scale culturomics to skin. Integrate cosmetic-firm strain libraries (L'Oréal Lactobio ~10,000 strains; COSMAX internal library) + JAX (Julia Oh) follow-up + Bay Area Microbiome Lab + S-Biomedic internal collection into a 1,000–5,000 culturable skin strain range with whole-genome sequencing + functional assay (AMP secretion, biofilm, host-cell signaling). The resulting dataset becomes training corpus for a function-prediction foundation model.
Operational measurement: 3 years culturomics + 2 years foundation-model training = 5-year timeline. Plausibly a 200–500 publication arc spanning academic and industry co-authorship.
Most plausible host: academic-industry hybrid. JAX (Julia Oh) + Stanford (David Relman) + 4–5 strain-library industry partners.
Bet 4 — A skin-gut axis cosmetic active (closes Gap 14)
Gap closed: Gap 14 — skin-gut-brain-axis claims outrun mechanistic evidence specifically for cosmetic endpoints.
Core of the bet: a ZOE-PREDICT-style [4] RCT testing whether systemic intervention (probiotic, prebiotic, postbiotic, or diet) on gut microbiome produces causal effects on skin phenotype (hydration, TEWL, wrinkle, pigmentation, age-spot) under an intervention design. 5-year timeline: year 1 cohort selection, years 2–3 intervention, years 4–5 follow-up.
Operational measurement: success is closing at least one mechanistic chain (e.g., gut Akkermansia → SCFA → systemic AHR → skin barrier function) causally. The book's view: the Takei-Hashimoto AHR axis [39] is the most cleanly groundtruthed mechanism, so dietary modulation of an AHR-target cosmetic active is the most plausible bet.
Most plausible host: a nutraceutical firm (ZOE, Viome) partnered with a cosmetic firm (L'Oréal, Unilever). The pharma route is cost-prohibitive.
Bet 5 — Time biology × microbiome (circadian skin cycles)
Gap closed: a new gap — circadian skin-microbiome cycles are mentioned (Frontiers Aging 2024 [14]) but mechanistically uninstrumented.
Core of the bet: characterize the circadian (24-hour) rhythm of skin microbiome and run an RCT testing whether cosmetic active application timing modulates efficacy. Endpoint: whether the same active yields different endpoint readouts depending on morning vs evening application. Operational measurement: 1 year baseline cycle characterization + 2 years timing-modulated intervention = 3-year timeline.
Most plausible host: academic first (Northwestern, Surrey, Penn — strong chronobiology groups) followed by cosmetic-firm partnership. The book's view: this bet is the most under-explored with the lowest cost and the most distinctive publication upside.

12.8 Closing — the marketing-vs-science delta and trust as competitive advantage
This book has measured the delta between science and marketing in the cosmetic industry. Unilever's 60% / 5–6→1–2 / 75% KPIs are plausibly true as engineering KPIs — internal process measurements. The implicit suggestion that virtual cohorts can substitute for efficacy and safety clinical trials is methodologically aggressive and externally unvalidated (Gap 15). L'Oréal's Skin Genius, COSMAX's Microbiome AI platform [10], Shiseido's Voyager [38], and POND'S in-store diagnostic [34] all share the same pattern — impressive operational engineering, thin auditable science.
AI hype has internal cycles (like most technology cycles). Cosmetic AI is currently traversing the inflated-expectations phase of 2023–2025 and approaching a 2026–2028 trough of disillusionment. Whether to prepare for the trough now or to deny it is a firm-level decision. The book's diagnosis: publication is the most stable asset through such a phase. Insilico's rentosertib readout in May 2025 [19] was the moment Insilico skipped the pharma-AI disillusionment phase — the publication itself was a competitive signal.
The cosmetic industry's equivalent moment is the first peer-reviewed clinical readout of an AI-designed cosmetic active, and whichever firm publishes it becomes the reference firm of the next five years. Trust as competitive advantage — the book's final claim. When, where, and which firm will reach the moment at which a publication strategy generates greater ROI than an IP-first strategy is a firm-level decision, but it is already visible as an industry signal.
The marketing language of skin microbiome balance will plausibly mature into strain-level + audited claim language around 2030. The separation between firms that lead and firms that get overtaken during that maturation is the industry narrative of the next five years.
12.9 Open questions for the next book
This section names what the book can answer and what it cannot. The five questions below are unanswerable in the May 2026 literature and define the gaps the next book — or the next edition — should close.
- Which firm, region, and endpoint will produce the first peer-reviewed clinical readout of an AI-designed cosmetic active? This is the direct closure of Gap 1. Book's wager: Korea (COSMAX-Dankook) or a mid-tier EU firm. Most likely endpoint: anti-aging (wrinkle volume) or anti-inflammatory (atopic-dermatitis cosmetic adjuvant).
- Under which regulatory framework will the first live engineered cosmetic microbe gain approval? Geltor-style GRAS extension, Seres-style drug route, or a newly created category. Book's wager: MFDS or EU CTR is world first; FDA's rulemaking cycle is too long.
- Which data coalition will train the first skin-microbiome foundation-model prototype? Unilever + L'Oréal + COSMAX, or an academic-government federated-learning consortium. Book's wager: academic-government federated learning leapfrogs an industry coalition but not before 2030.
- Will Amorepacific and LG H&H adopt a publication strategy, or will they explicitly formalize an IP-first strategy? Closure of Gap 4. Book's wager: when competitive pressure crosses a threshold (e.g., COSMAX or an EU firm publishes an AI-designed cosmetic active), a pivot to publication follows — but unlikely visible before 2027.
- Which mechanistic chain will produce the first causal evidence for a skin-gut axis cosmetic active? Closure of Gap 14. Book's wager: the AHR axis (Takei-Hashimoto lineage [39]) is the cleanest ground truth, so AHR-targeting dietary modulation × skin barrier is the most plausible clinical chain.
When these five questions close — plausibly around 2030 — the frame for the next book will differ. The post-inflection microbiome × AI cosmetic industry will already have entered its maturation phase, and the book you are reading in May 2026 records the second inflection point — the first was 2021 [21], the second is whichever year the first peer-reviewed clinical readout of an AI-designed cosmetic active is published.
Until that day fundamental research is not obsolete. After that day it will still not be.
References
- Abramson, J., Adler, J., Dunger, J. et al. (2024). Accurate structure prediction of biomolecular interactions with AlphaFold 3. Nature 630, 493–500.
- Amorepacific (2024). Amorepacific R&D — postbiotic and microbiome research disclosures. Amorepacific press, 2024. [Amorepacific, 2024]
- Arcaea (2024). Arcaea — precision prebiotics for the skin microbiome. Arcaea product disclosures, 2024. [Arcaea, 2024]
- Asnicar, F., Berry, S. E., Valdes, A. M. et al. (2021). Microbiome connections with host metabolism and habitual diet from 1,098 deeply phenotyped individuals (ZOE PREDICT 1). Nature Medicine 27, 321–332.
- Atallah, C., Charcosset, C. et al. (2025). Bioengineered Skin Microbiome: Advances in Cosmetic Applications, Strain-Level Resolution, and Regulatory Considerations. Cosmetics 12(5):205.
- Baek, M., DiMaio, F., Anishchenko, I. et al. (2021). Accurate prediction of protein structures and interactions using a three-track neural network (RoseTTAFold). Science 373, 871–876.
- Brödel, A. K., Charpentier, E. et al. (2024). In situ base editing of microbiome communities. Nature 632, 633–641.
- Carrieri, A. P., Haiminen, N., Maudsley-Barton, S. et al. (2021). Explainable AI reveals changes in skin microbiome composition linked to phenotypic differences. Scientific Reports 11:4565.
- Chai Discovery (2024). Chai-1: Decoding the molecular interactions of life. Chai Discovery technical report, 2024. [Chai Discovery, 2024]
- COSMAX USA (2024). COSMAX unveils 2nd-Generation Skin Microbiome platform + Microbiome AI. Global Cosmetic Industry, 2024. [COSMAX, 2024]
- Di Guardo, A., Trovato, F., Cantisani, C. et al. (2025). Artificial Intelligence in Cosmetic Formulation: Predictive Modeling for Safety, Tolerability, and Regulatory Perspectives. Cosmetics 12(4):157.
- Eligo Bioscience (2023). European patent EP4240843A1 — In situ CRISPR base editing for skin microbiome modulation. Eligo patent disclosure, 2023. [Eligo, 2023]
- Fitzgibbon, S., Tomida, S., Chiu, B.-H. et al. (2013). Propionibacterium acnes strain populations in the human skin microbiome associated with acne36325-9/fulltext). Journal of Investigative Dermatology 133, 2152–2160.
- Frontiers Aging (2024). Frontiers in Aging — skin barrier and microbiome circadian dynamics review. Frontiers in Aging, 2024. [Frontiers Aging, 2024]
- Geltor (2025). FDA issues GRAS "no questions" letter for Geltor's PrimaColl bioengineered collagen. Geltor press, Oct 2025. [Geltor, 2025]
- Hashimoto, R., Kawano, K. et al. (2022). Galactomyces ferment filtrate activates NRF2-mediated antioxidant signaling in keratinocytes. Journal of Dermatological Science, 2022. [Hashimoto et al., 2022]
- Haykal, D., Flament, F., Amar, D. et al. (2025). Cosmetogenomics unveiled: a systematic review of AI, genomics, and the future of personalized skincare. Frontiers in Artificial Intelligence 8:1660356.
- Hayes, T., Rao, R., Akin, H. et al. (2025). Simulating 500 million years of evolution with a language model (ESM3). Science 387, 850–858.
- Insilico Medicine (Ren, F., Zhavoronkov, A. et al.) (2025). A generative AI-discovered TNIK inhibitor for idiopathic pulmonary fibrosis: a randomized phase 2a trial. Nature Medicine, May 2025. [Insilico Medicine, 2025]
- Ji, Y., Zhou, Z., Liu, H., Davuluri, R. V. (2021). DNABERT: pre-trained Bidirectional Encoder Representations from Transformers model for DNA-language in genome. Bioinformatics 37, 2112–2120.
- Jumper, J., Evans, R., Pritzel, A. et al. (2021). Highly accurate protein structure prediction with AlphaFold. Nature 596, 583–589.
- Kim, M.-S., Mun, S. et al. (2021). Spermidine-induced recovery of human dermal structure and barrier function. Communications Biology 4:71.
- Kim, B.-S., Choi, S. et al. (2023). Clinical efficacy of EPI-7 postbiotic in dermal aging — a randomized controlled trial. International Journal of Molecular Sciences 24(5):4634.
- Krishna, R., Wang, J., Ahern, W. et al. (2024). Generalized biomolecular modeling and design with RoseTTAFold All-Atom. Science 384, eadl2528.
- Lagier, J.-C., Dubourg, G., Million, M. et al. (2018). Culturing the human microbiota and culturomics. Nature Reviews Microbiology 16, 540–550.
- LG H&H (2025). LG Household & Health Care — oral microbiome research MOU disclosure. LG H&H press, 2025. [LG H&H, 2025]
- Li, Z., Xia, J., Jiang, L. et al. (2021). Characterization of the human skin resistome and identification of two microbiota cutotypes (iHSMGC). Microbiome 9:47.
- Mun, S., Kim, M.-S. et al. (2025). FACE-LINK: A 950-subject Korean facial skin microbiome cohort for personalized cosmetic development. Frontiers in Cellular and Infection Microbiology, 2025. [Mun et al., 2025]
- Nakatsuji, T., Chen, T. H., Narala, S. et al. (2017). Antimicrobials from human skin commensal bacteria protect against Staphylococcus aureus. Science Translational Medicine 9, eaah4680.
- Nakatsuji, T., Hata, T. R., Tong, Y. et al. (2021). Development of a human skin commensal microbe for bacteriotherapy of atopic dermatitis (ShA9 Phase 1). Nature Medicine 27, 700–709.
- Papoutsoglou, G., Tarazona, S., Lopes, M. B. et al. (2023). Machine learning approaches in microbiome research: challenges and best practices. Frontiers in Microbiology 14:1261889.
- Parallel Health (2024). Parallel Health — phage cocktail for skin microbiome modulation. Parallel Health product disclosures, 2024. [Parallel Health, 2024]
- Pfizer (2025). Pfizer × XtalPi AI partnership expansion. Pfizer press, 2025. [Pfizer, 2025]
- POND'S (Unilever) (2024). POND's Skin Institute microbiome analyzer — 60-minute in-store consumer device. Unilever press, May 2024. [POND'S, 2024]
- Ren, F., Aliper, A., Chen, J. et al. (2024). A small-molecule TNIK inhibitor targets fibrosis in preclinical and clinical models (rentosertib). Nature Biotechnology 42, 1158–1167.
- S-Biomedic (2024). S-Biomedic — engineered C. acnes expressing RoxP for sebum redox modulation. S-Biomedic disclosure, 2024. [S-Biomedic, 2024]
- Seres Therapeutics (2024). VOWST (SER-109) — first FDA-approved oral microbiome therapeutic commercial update. Seres Therapeutics press, 2024. [Seres Therapeutics, 2024]
- Shiseido + Accenture (2024). Shiseido develops AI systems for ingredient biodegradability and the Voyager formulation platform. Global Cosmetics News, Feb 2024. [Shiseido + Accenture, 2024]
- Takei, K., Mitoma, C. et al. (2015). Galactomyces ferment filtrate as an aryl hydrocarbon receptor agonist restoring filaggrin expression. Experimental Dermatology 24, 686–691.
- Unilever Beauty & Wellbeing R&D (2025). How Unilever's pioneering skin microbiome research is shaping product innovation. Unilever news, 2025. [Unilever, 2025]
- Unilever Beauty & Wellbeing (2025). SXSW 2025 — AI/ML/data behind Unilever's latest launches. Unilever news, Mar 2025. [Unilever, 2025-SXSW]
- Wohlwend, J., Corso, G., Passaro, S. et al. (2024). Boltz-1: Democratizing biomolecular interaction modeling. bioRxiv, 2024. [Wohlwend et al., 2024]
- ZOE Health (2024). ZOE PREDICT — nutritional intervention based on microbiome and metabolic phenotyping. Nature Medicine, 2024. [ZOE, 2024]
- Novo Nordisk (2024). Novo Nordisk × Cradle — generative protein design partnership announcement. Novo Nordisk press, Nov 2024. [Novo Nordisk, 2024]